MANUFACTURER
MANUFACTURER
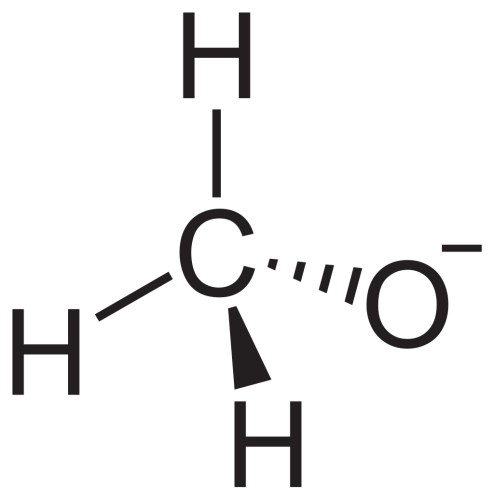
Potassium Methoxide
Regular price:
Regular price:
product unavailable
 add to wish list
add to wish list
 Description
Description
Potassium Methoxide
Other Names: Potassium methylate, Methanolate (potassium salt), Methanolate de potassium
CAS Number: 865-33-8
Sum Formula: CH₃KO
Molar Mass: 70.13 g/mol
Appearance: White to off-white crystalline solid or powder (often handled as a solution in methanol)
Potassium methoxide is a strong, non-nucleophilic base and a potassium alkoxide. It is commonly used in organic synthesis and industrial processes that require strong basic conditions, such as transesterification and condensation reactions. It is highly reactive with moisture and must be handled under anhydrous conditions.
Chemical and Physical Properties
Density: Approx. 1.4 g/cm³ (solid, estimated)
Melting Point: ~170°C (decomposes)
Boiling Point: Not applicable (decomposes)
Solubility in Water: Reacts violently
Solubility in Other Solvents: Soluble in methanol and ethanol; reacts with moisture
Odor: Pungent, methanol-like when dissolved
Applications
Organic Synthesis: Used as a strong base for deprotonation and E2 elimination reactions.
Biodiesel Production: Catalyst for transesterification of triglycerides to fatty acid methyl esters.
Pharmaceutical and Agrochemical Synthesis: Involved in condensation and methylation reactions.
Polymer Chemistry: Utilized in polymer modification and as an initiator in certain polymerizations.
Safety



Signal Word: Danger
H228 (50.6%): Flammable solid [Danger Flammable solids]
H251 (97.9%): Self-heating; may catch fire [Danger Self-heating substances and mixtures]
H302 (53.6%): Harmful if swallowed [Warning Acute toxicity, oral]
H314 (99.6%): Causes severe skin burns and eye damage [Danger Skin corrosion/irritation]
H318 (53.6%): Causes serious eye damage [Danger Serious eye damage/eye irritation]
P210, P235, P240, P241, P260, P264, P264+P265, P270, P280, P301+P317, P301+P330+P331, P302+P361+P354, P304+P340, P305+P354+P338, P316, P317, P321, P330, P363, P370+P378, P405, P407, P410, P413, P420, and P501
Handling and Storage
Storage Conditions: Store in a tightly sealed container under inert atmosphere (e.g., nitrogen or argon), in a cool, dry, and well-ventilated area. Avoid exposure to air and moisture.
Handling Precautions: Handle in a glovebox or under anhydrous conditions. Avoid any contact with water or humid air. Use appropriate PPE and non-sparking tools.