MANUFACTURER
MANUFACTURER
Heterocycles are cyclic compounds in which one or more ring atoms are heteroatoms—most commonly N, O, or S—rather than carbon. They are ubiquitous in natural products, pharmaceuticals, and materials, and their heteroatoms strongly influence ring electronics and reactivity.
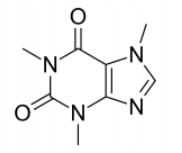
1-Ethyltheobromine (Caffeine Analogue) Spectroscopically Pure

Availability: Large quantity
Dispatched within: 10 days
1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 1000ml
![1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 1000ml](/environment/cache/images/productGfx_16443_200_200/2025-05-16_14h48_25.png?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 100ml
![1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 100ml](/environment/cache/images/productGfx_678_200_200/9d41600afcc67a3e453202b22f47ab6f.jpg?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 10ml
![1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 10ml](/environment/cache/images/productGfx_16439_200_200/2025-05-16_14h48_25.png?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 500ml
![1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 500ml](/environment/cache/images/productGfx_16441_200_200/2025-05-16_14h48_25.png?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 5000ml
![1,4-Dioxane ( Dioxane : 1,4-Dioxacyclohexane p-Dioxane : [6]-crown-2 : Diethylene dioxide - 5000ml](/environment/cache/images/productGfx_16445_200_200/2025-05-16_14h48_25.png?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
1-Hydroxybenzotriazole

⚠️ This product can only be purchased by registered business or government entities.
✉️ Declaration of use is required.
Availability: na wyczerpaniu
Dispatched within: 5 days
2,2′-Bipyridine : Bipyridyl : Dipyridyl : Bipy : Bpy : Dipy : 2,2-Bipyridyl - Chelating - 250g

Availability: Large quantity
Dispatched within: 5 days
Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 100g
![Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 100g](/environment/cache/images/productGfx_824_200_200/1f59078390457ba11926d197e8b8927e.jpg?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 10g
![Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 10g](/environment/cache/images/productGfx_827_200_200/0682b4f0ea01b4cb60f1f4d895f9807b.jpg?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 250g
![Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 250g](/environment/cache/images/productGfx_823_200_200/8110ff21f5a7123cb8103de32d0afe8a.jpg?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 25g
![Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 25g](/environment/cache/images/productGfx_826_200_200/4efe3c983d29dc956a3dab72209ee949.jpg?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 50g
![Antipyrine ( Phenazone - Antipyrin - Phenazon ) [CAS: 60-80-0] - 50g](/environment/cache/images/productGfx_825_200_200/ec671515ae7f722e1e0cffbea47f04ad.jpg?overlay=1)
Availability: Large quantity
Dispatched within: 5 days
Caffeine Powder Anhydrous Fluffy 99,9% - 100g

Availability: Large quantity
Dispatched within: 5 days
Caffeine Powder Anhydrous Fluffy 99,9% - 10kg = 10 000g
Availability: Large quantity
Dispatched within: 5 days
Caffeine Powder Anhydrous Fluffy 99,9% - 500g
Availability: Large quantity
Dispatched within: 5 days
Caffeine Powder Anhydrous Fluffy 99,9% 1kg - 1kg = 1000g
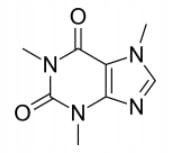
Availability: Large quantity
Dispatched within: 5 days
Pyridine Pure >99% - 1000ml
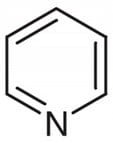
✈︎ Shipping Limitations: We do not ship this product to: US | Unless government approved ⓘ
Availability: Large quantity
Dispatched within: 5 days
Pyridine Pure >99% - 100ml
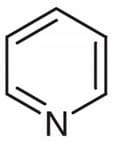
✈︎ Shipping Limitations: We do not ship this product to: US | Unless government approved ⓘ
Availability: Large quantity
Dispatched within: 5 days
Pyridine Pure >99% - 500ml
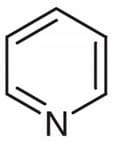
✈︎ Shipping Limitations: We do not ship this product to: US | Unless government approved ⓘ
Availability: Large quantity
Dispatched within: 5 days
Tetrahydrofurane ( THF ) Oxolane Analytically Pure
Availability: Large quantity
Dispatched within: 5 days
Heterocycles are classified by ring size (typically 3–7 members; larger rings called macrocycles), degree of saturation (saturated, partially unsaturated, aromatic), and the identity/number/position of heteroatoms. Aromatic heterocycles obey Hückel’s 4n+2 π-electron rule and can be π-excessive (electron-rich; e.g., pyrrole, furan, thiophene) or π-deficient (electron-poor; e.g., pyridine, diazines) depending on whether heteroatom lone pairs contribute to the aromatic sextet. This distinction governs both regioselectivity and mechanism in substitution reactions.
Electronic structure and basicity.
Nitrogen-containing heterocycles show wide basicity variation. In pyridine-type rings, the N lone pair lies in an sp² orbital orthogonal to the π system, making it available for protonation and coordination (relatively basic). In pyrrole-type rings, the lone pair is part of the aromatic π system, so protonation would disrupt aromaticity (much less basic). Additional heteroatoms and ring fusion further tune pKₐ, dipole moments, and hydrogen-bonding profiles—key parameters in drug design.
Reactivity patterns.
-
Electron-rich aromatic heterocycles undergo electrophilic aromatic substitution (EAS) readily, often at positions that best stabilize the σ-complex (e.g., C-2 in pyrrole/furan/thiophene). They are also prone to oxidation.
-
Electron-poor aromatic heterocycles are deactivated toward EAS but activated toward nucleophilic aromatic substitution (S_NAr) and metal-catalyzed cross-couplings.
-
Saturated heterocycles behave like functionalized alicycles; their heteroatoms enable substitutions, oxidations, and ring-opening reactions.
Synthesis.
Heterocycles are commonly built via cyclization strategies that form C–X and/or C–C bonds in one step. Representative methods include:
-
Paal–Knorr syntheses (five-membered N/O/S heteroaromatics from 1,4-dicarbonyls).
-
Fischer indole synthesis (indoles from arylhydrazones).
-
Hantzsch and Biginelli reactions (dihydropyridines, pyrimidinones).
-
Cycloadditions (e.g., azide–alkyne to triazoles).
-
Ring-closing amidation/esterification for lactams and lactones.
Modern routes often use Pd/Ni catalysis or photoredox methods to assemble densely substituted heterocycles.
Importance and examples.
Heterocycles are central scaffolds in biology (nucleobases, sugars, cofactors) and medicine (e.g., β-lactams, azoles, quinolines). Their ability to present lone pairs, dipoles, and defined 3D shapes makes them ideal for binding to enzymes and receptors, and for tuning solubility, permeability, and metabolic stability in functional molecules.