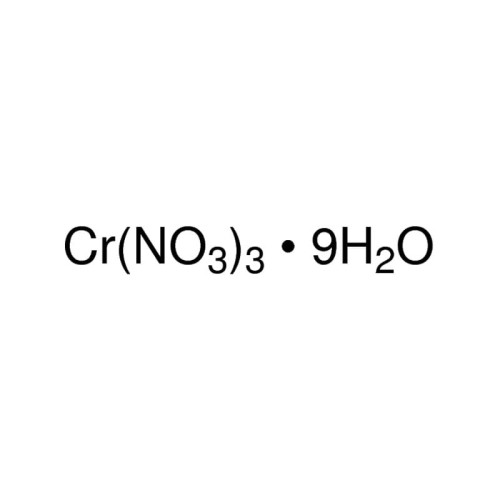
Chromium (III) nitrate nonahydrate
Regular price:
Regular price:
product unavailable
 add to wish list
add to wish list
 Description
Description
Chromium(III) Nitrate Nonahydrate
Other Names: Chromium nitrate nonahydrate, Chromic nitrate nonahydrate
CAS Number: 7789-02-8
Sum Formula: Cr(NO₃)₃·9H₂O
Molar Mass: 400.15 g/mol
Appearance: Dark purple to violet crystalline solid
Chromium(III) nitrate nonahydrate is an inorganic salt of trivalent chromium and nitric acid, commonly encountered as a hydrated crystalline compound. It is primarily used in laboratories and industry for synthesis, dyeing, and as a catalyst precursor.
Chemical and Physical Properties
Density: 1.85 g/cm³
Melting Point: 60°C (decomposes on further heating)
Boiling Point: Not applicable (decomposes)
Solubility in Water: Soluble
Solubility in Other Solvents: Slightly soluble in alcohol; insoluble in nonpolar solvents
Odor: Odorless
Applications
Inorganic Synthesis: Used to produce other chromium(III) compounds.
Catalysis: Precursor to chromium-based catalysts.
Textile and Leather Industry: Sometimes applied in dyeing or tanning processes.
Analytical Chemistry: Reagent in qualitative and quantitative analysis involving chromium species.
Safety

Signal Word: Warning
H272 (93.2%): May intensify fire; oxidizer [Danger Oxidizing liquids; Oxidizing solids]
H302 (15.7%): Harmful if swallowed [Warning Acute toxicity, oral]
H315 (42.9%): Causes skin irritation [Warning Skin corrosion/irritation]
H317 (55%): May cause an allergic skin reaction [Warning Sensitization, Skin]
H319 (45%): Causes serious eye irritation [Warning Serious eye damage/eye irritation]
H332 (57.1%): Harmful if inhaled [Warning Acute toxicity, inhalation]
H411 (55%): Toxic to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard]
P210, P220, P261, P264, P264+P265, P270, P271, P272, P273, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P317, P321, P330, P332+P317, P333+P317, P337+P317, P362+P364, P370+P378, P391, and P501
Handling and Storage
Storage Conditions: Store in a tightly closed container in a cool, dry, and well-ventilated area. Protect from heat, moisture, and incompatible substances such as strong oxidizing or reducing agents.
Handling Precautions: Avoid generating dust and contact with skin or eyes. Use appropriate protective equipment and good laboratory practices.