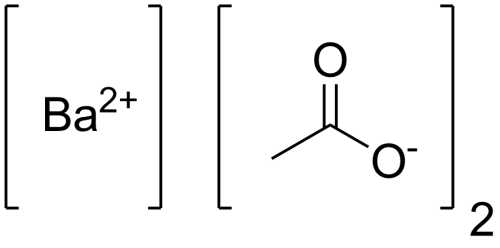
Barium Acetate
Regular price:
Regular price:
product unavailable
 add to wish list
add to wish list
 Description
Description
Barium Acetate
Other Names: Acetic acid, barium salt; Barium diacetate
CAS Number: 543-80-6
Sum Formula: Ba(C₂H₃O₂)₂
Molar Mass: 255.42 g/mol
Appearance: White crystalline solid or powder
Barium acetate is an inorganic salt formed by the reaction of barium carbonate or barium hydroxide with acetic acid. It is used in the production of other barium compounds, in chemical synthesis, and in various industrial processes including printing and dyeing.
Chemical and Physical Properties
Density: 2.47 g/cm³
Melting Point: 450°C (decomposes)
Boiling Point: Not applicable (decomposes)
Solubility in Water: Soluble (55 g/100 mL at 0°C)
Solubility in Other Solvents: Slightly soluble in alcohol; insoluble in nonpolar solvents
Odor: Odorless
Applications
Chemical Manufacturing: Used in the preparation of other barium salts.
Textile and Dye Industry: Employed as a mordant in dyeing processes.
Laboratory Reagent: Used in analytical chemistry and chemical synthesis.
Lubricants and Greases: May be used in formulation of certain high-temperature lubricants.
Safety

Signal Word: Danger
H302 (99.7%): Harmful if swallowed [Warning Acute toxicity, oral]
H332 (99.7%): Harmful if inhaled [Warning Acute toxicity, inhalation]
P261, P264, P270, P271, P301+P317, P304+P340, P317, P330, and P501
Handling and Storage
Storage Conditions: Store in a tightly closed container in a dry, well-ventilated area. Protect from moisture and incompatible substances such as strong acids and oxidizing agents.
Handling Precautions: Avoid generating dust. Do not breathe dust or ingest the material. Use suitable personal protective equipment and handle in accordance with good laboratory practices.