Chelation involves ligands (chelators) that possess multiple Lewis-basic donor sites—commonly N, O, or S atoms—capable of simultaneously coordinating a single metal center. When these donors bind, they generate chelate rings (often 5- or 6-membered), producing complexes denoted, for example, as [M(L)]ⁿ where L is multidentate. Ligands are classified by denticity (bidentate, tridentate, tetradentate, etc.); classic examples include ethylenediamine (en), oxalate, and EDTA (hexadentate).
Chelate effect and thermodynamics.
Chelated complexes are usually more stable than those formed by comparable monodentate ligands, a phenomenon known as the chelate effect. The dominant driver is entropy: replacing several monodentate ligands with one multidentate ligand increases the number of free particles in solution upon complex formation, making ΔS more positive and thus ΔG more negative. Enthalpic contributions (stronger binding due to preorganization or reduced solvent competition) can reinforce this, but the effect is mainly entropic.
Structure and bonding.
Chelates can adopt geometries matching the metal’s preferred coordination number and electronic configuration (octahedral, square planar, tetrahedral, etc.). Ligand field strength influences high-spin vs low-spin states, magnetic properties, and electronic spectra. Ring size, rigidity, and bite angle (donor–metal–donor angle) strongly affect stability and reactivity; overly strained rings or mismatched bite angles destabilize the complex.
Kinetics and reactivity.
Chelation often slows ligand dissociation, giving kinetic inertness in addition to thermodynamic stability—especially for low-spin d⁶ metals (Co³⁺, Ru²⁺) and square-planar d⁸ metals (Pt²⁺). Chelates participate in substitution, redox, and catalytic cycles; fine-tuning donor atoms and geometry is a key strategy in homogeneous catalysis.
Applications.
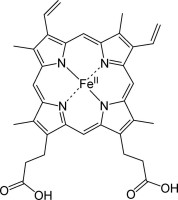
Chelates are central in biology (heme–Fe, chlorophyll–Mg, metalloproteins), medicine (EDTA in metal poisoning; Gd-chelates in MRI; Pt-chelates in chemotherapy), analytical chemistry (complexometric titrations), and industry (water softening, metal separation). Their ability to selectively bind metals with high stability and controllable kinetics makes them indispensable across chemical and biochemical systems.