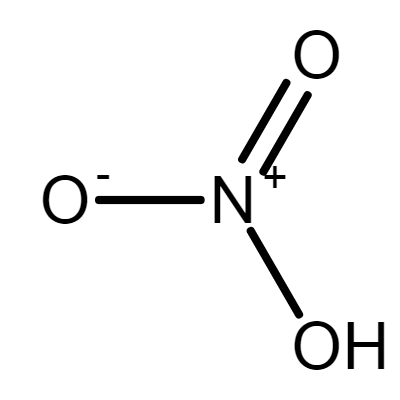
NITRIC ACID
Regular price:
Regular price:
product unavailable
 add to wish list
add to wish list




 0
0
 Description
Description
Nitric Acid
Other Names: Aqua fortis, Hydrogen nitrate
CAS Number: 7697-37-2
Sum Formula: HNO₃
Molar Mass: 63.01 g/mol
Appearance: Colorless to yellowish liquid (fuming in moist air)
Nitric acid is a highly corrosive and strong mineral acid. It is commonly used as a reagent in chemical synthesis, metal processing, and nitration reactions. Fuming forms contain dissolved nitrogen oxides and appear yellow to red-brown.
1. This product is available to registered business and government entities only. Company details and tax number must be provided at checkout. Orders without this information will be cancelled.
2. This product requires a declaration of intended use to be completed before the order can be processed. A declaration form template can be found at the bottom of the product description. Please send the completed declaration to synthetika@synthetikaeu.com .
Chemical and Physical Properties
Density: 1.51 g/cm³ (concentrated, ~68%)
Melting Point: -42°C
Boiling Point: 83°C (concentrated solution)
Solubility in Water: Completely miscible
Solubility in Other Solvents: Miscible with polar solvents like methanol and acetone
Odor: Pungent, suffocating
pH: <1 (strong acid)
Applications
-
Chemical Manufacturing: Used in nitration of organic compounds, production of fertilizers, explosives, and dyes.
-
Metal Treatment: Employed in etching, pickling, and passivation of stainless steel.
-
Laboratory Reagent: Commonly used in acid digestion and quantitative analysis procedures.
-
Environmental Technology: Involved in NOx scrubbing and pollution control processes.
Safety



Signal Word: Danger
H272 (98.6%): May intensify fire; oxidizer [Danger Oxidizing liquids; Oxidizing solids]
H290 (18.9%): May be corrosive to metals [Warning Corrosive to Metals]
H314 (> 99.9%): Causes severe skin burns and eye damage [Danger Skin corrosion/irritation]
H318 (17.5%): Causes serious eye damage [Danger Serious eye damage/eye irritation]
H331 (11.9%): Toxic if inhaled [Danger Acute toxicity, inhalation]
P210, P220, P234, P260, P261, P264, P264+P265, P271, P280, P301+P330+P331, P302+P361+P354, P304+P340, P305+P354+P338, P316, P317, P321, P363, P370+P378, P390, P403+P233, P405, P406, and P501
First Aid Measures
Eye Contact: Rinse immediately with plenty of water for at least 15 minutes. Seek urgent medical attention.
Skin Contact: Remove contaminated clothing. Rinse skin thoroughly with water. Seek medical attention.
Inhalation: Move to fresh air. Administer oxygen if needed. Seek immediate medical help.
Ingestion: Do not induce vomiting. Rinse mouth. Get medical attention immediately.
Handling and Storage
Storage Conditions: Store in a corrosion-resistant container with a resistant inner liner. Keep in a cool, well-ventilated area away from incompatible materials such as organic substances, bases, and reducing agents.
Handling Precautions: Use only in a chemical fume hood. Avoid all contact with skin, eyes, and clothing. Use full PPE and acid-resistant materials.