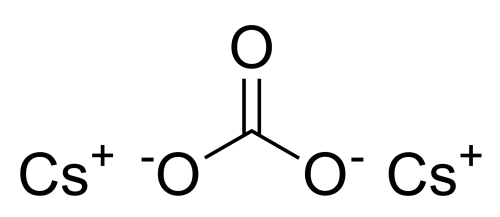
Cesium carbonate
Regular price:
Regular price:
product unavailable
 add to wish list
add to wish list
 Description
Description
Cesium Carbonate
Other Names: Dicaesium carbonate, Caesium carbonate
CAS Number: 534-17-8
Sum Formula: Cs₂CO₃
Molar Mass: 325.82 g/mol
Appearance: White hygroscopic powder
Cesium carbonate is an inorganic salt of cesium and carbonic acid, appearing as a white, water-soluble powder. It is commonly used in organic synthesis as a strong, non-nucleophilic base and is valued in catalytic processes due to its high basicity and solubility in polar solvents.
Chemical and Physical Properties
Density: ~4.07 g/cm³
Melting Point: Decomposes before melting
Solubility in Water: Very soluble, forms an alkaline solution
Solubility in Other Solvents: Slightly soluble in alcohols
Odor: Odorless
Applications
Organic Synthesis: Functions as a strong base in a variety of organic reactions, including alkylation, esterification, and cross-coupling reactions.
Catalysis: Utilized in phase-transfer catalysis and palladium-catalyzed coupling reactions.
Material Science: Used in the preparation of optoelectronic materials and as a dopant in electronic applications.
Inorganic Synthesis: Employed in the preparation of other cesium-containing compounds.
Safety

Signal Word: Danger
H315 (41.5%): Causes skin irritation [Warning Skin corrosion/irritation]
H318 (59%): Causes serious eye damage [Danger Serious eye damage/eye irritation]
H319 (40%): Causes serious eye irritation [Warning Serious eye damage/eye irritation]
H335 (41%): May cause respiratory irritation [Warning Specific target organ toxicity, single exposure; Respiratory tract irritation]
H361f (54.5%): Suspected of damaging fertility [Warning Reproductive toxicity]
H373 (58.5%): May causes damage to organs through prolonged or repeated exposure [Warning Specific target organ toxicity, repeated exposure]
P203, P260, P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P305+P354+P338, P317, P318, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501
Handling and Storage
Storage Conditions: Store in a tightly closed container in a dry, cool, and well-ventilated area. Avoid contact with moisture and strong acids.
Handling Precautions: Avoid dust formation. Use appropriate protective equipment to prevent contact with skin and eyes.