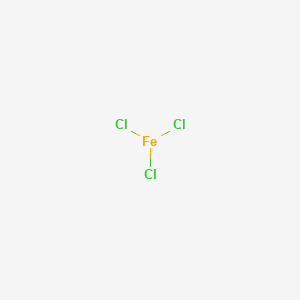
Iron (III) Chloride, Anhydrous
Regular price:
Regular price:
product unavailable
 add to wish list
add to wish list




 0
0
 Description
Description
Iron (III) Chloride, Anhydrous
Other Names: Ferric chloride, Iron trichloride
CAS Number: 7705-08-0
Sum Formula: FeCl₃
Molar Mass: 162.20 g/mol
Appearance: Dark brown to black crystalline solid
Iron (III) chloride anhydrous is a highly hygroscopic, corrosive inorganic compound commonly used as a Lewis acid catalyst, etching agent, and oxidizing agent in various industrial and laboratory processes.
Chemical and Physical Properties
Density: 2.90 g/cm³ (20°C)
Melting Point: 306°C
Boiling Point: ~316°C (decomposes)
Solubility in Water: Reacts exothermically with water, forming hydrochloric acid and ferric hydroxides
Solubility in Organic Solvents: Slightly soluble in ethanol and acetone
Odor: Strong hydrogen chloride (acidic) odor when hydrolyzed
Stability: Hygroscopic; reacts with moisture and releases HCl fumes
Applications
Used as a catalyst in Friedel-Crafts and other organic synthesis reactions
Applied as an etching agent for copper and other metals (e.g., in PCB manufacturing)
Used in wastewater treatment as a coagulant (technical grade)
Serves as an oxidizing agent in chemical processes
Applied in pigment and dye manufacturing and surface treatments
Safety

Signal Word: Danger
H302 (96.6%): Harmful if swallowed [Warning Acute toxicity, oral]
H314 (75.4%): Causes severe skin burns and eye damage [Danger Skin corrosion/irritation]
H315 (24.4%): Causes skin irritation [Warning Skin corrosion/irritation]
H318 (24.5%): Causes serious eye damage [Danger Serious eye damage/eye irritation]
H412 (72.4%): Harmful to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard]
First Aid Measures
Eye Contact: Rinse immediately with water for at least 15 minutes. Seek urgent medical attention
Skin Contact: Remove contaminated clothing and wash thoroughly with water and soap
Inhalation: Move to fresh air. Seek medical help if symptoms develop
Ingestion: Do not induce vomiting. Rinse mouth and seek medical attention immediately
Handling and Storage
Store in a cool, dry, and tightly sealed container
Protect from moisture and humidity to prevent hydrolysis and HCl release
Avoid contact with bases, strong oxidizers, and metals
Use under fume hood with appropriate PPE