Producenci
Producenci
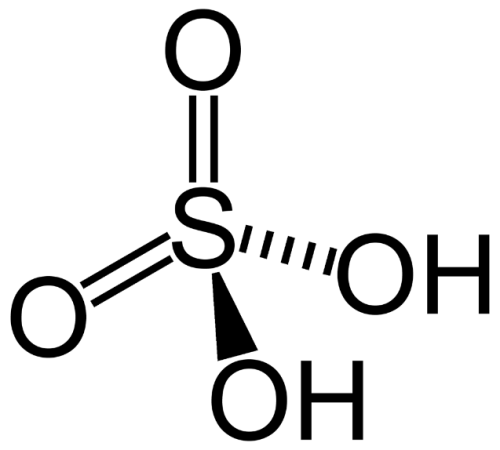
Kwas siarkowy ( Sulfuric Acid ) 96-98%
Cena regularna:
Cena regularna:
towar niedostępny
 dodaj do przechowalni
dodaj do przechowalni
 Opis
Opis
Sulfuric(VI) Acid 96–98%
Other Names: Oil of vitriol, Hydrogen sulfate
CAS Number: 7664-93-9
Sum Formula: H₂SO₄
Molar Mass: 98.08 g/mol
Appearance: Colorless to slightly yellow, dense, oily liquid
Sulfuric(VI) acid is a highly corrosive strong mineral acid composed of sulfur, oxygen, and hydrogen. It is one of the most important industrial chemicals, used in a wide range of applications, from chemical manufacturing to materials processing and environmental engineering. The concentrated form (96–98%) is particularly reactive and hygroscopic.
Chemical and Physical Properties
Density: 1.84 g/cm³ (at 20°C)
Melting Point: 10°C
Boiling Point: 337°C (decomposes partially)
Solubility in Water: Completely miscible with water (exothermic reaction)
Solubility in Other Solvents: Miscible with ethanol and methanol; reacts with many organic solvents
Odor: Odorless (may release choking fumes when decomposing)
Applications
Chemical Manufacturing: Used in the production of fertilizers, explosives, dyes, and detergents.
Petroleum Industry: Employed in refinery processes such as alkylation.
Batteries: Serves as the electrolyte in lead-acid batteries.
Surface Treatment: Used for metal pickling, cleaning, and electroplating preparation.
Laboratory Use: Acts as a strong acid catalyst, dehydrating agent, and analytical reagent.
Safety

Signal Word: Danger
P260, P264, P280, P301+P330+P331, P302+P361+P354, P304+P340, P305+P354+P338, P316, P321, P363, P405, and P501
Handling and Storage
Storage Conditions: Store in a tightly closed, corrosion-resistant container in a cool, dry, and well-ventilated area. Protect from moisture and incompatible materials such as organic substances, bases, and reducing agents.
Handling Precautions: Add acid to water, never the reverse. Use full PPE and conduct work in a fume hood or well-ventilated area.
Attention!
Sales Restrictions: Sales and transport of concentrated sulfuric acid are regulated in many countries due to its potential for misuse. Authorization, licensing, or end-user declarations may be required.