Producenci
Producenci
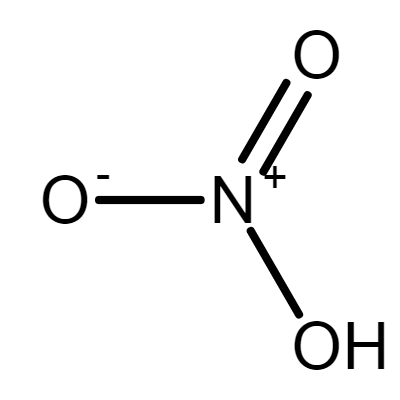
Kwas Azotowy 65%
Cena regularna:
Cena regularna:
towar niedostępny
 dodaj do przechowalni
dodaj do przechowalni




 0
0
 Opis
Opis
Nitric Acid
Other Names: Aqua fortis, Hydrogen nitrate
CAS Number: 7697-37-2
Sum Formula: HNO₃
Molar Mass: 63.01 g/mol
Appearance: Colorless to yellowish liquid (fuming in moist air)
Nitric acid is a highly corrosive and strong mineral acid. It is commonly used as a reagent in chemical synthesis, metal processing, and nitration reactions. Fuming forms contain dissolved nitrogen oxides and appear yellow to red-brown.
Chemical and Physical Properties
Density: 1.51 g/cm³ (concentrated, ~68%)
Melting Point: -42°C
Boiling Point: 83°C (concentrated solution)
Solubility in Water: Completely miscible
Solubility in Other Solvents: Miscible with polar solvents like methanol and acetone
Odor: Pungent, suffocating
pH: <1 (strong acid)
Applications
-
Chemical Manufacturing: Used in nitration of organic compounds, production of fertilizers, explosives, and dyes.
-
Metal Treatment: Employed in etching, pickling, and passivation of stainless steel.
-
Laboratory Reagent: Commonly used in acid digestion and quantitative analysis procedures.
-
Environmental Technology: Involved in NOx scrubbing and pollution control processes.
Safety



Signal Word: Danger
H272 (98.6%): May intensify fire; oxidizer [Danger Oxidizing liquids; Oxidizing solids]
H290 (18.9%): May be corrosive to metals [Warning Corrosive to Metals]
H314 (> 99.9%): Causes severe skin burns and eye damage [Danger Skin corrosion/irritation]
H318 (17.5%): Causes serious eye damage [Danger Serious eye damage/eye irritation]
H331 (11.9%): Toxic if inhaled [Danger Acute toxicity, inhalation]
P210, P220, P234, P260, P261, P264, P264+P265, P271, P280, P301+P330+P331, P302+P361+P354, P304+P340, P305+P354+P338, P316, P317, P321, P363, P370+P378, P390, P403+P233, P405, P406, and P501
First Aid Measures
Eye Contact: Rinse immediately with plenty of water for at least 15 minutes. Seek urgent medical attention.
Skin Contact: Remove contaminated clothing. Rinse skin thoroughly with water. Seek medical attention.
Inhalation: Move to fresh air. Administer oxygen if needed. Seek immediate medical help.
Ingestion: Do not induce vomiting. Rinse mouth. Get medical attention immediately.
Handling and Storage
Storage Conditions: Store in a corrosion-resistant container with a resistant inner liner. Keep in a cool, well-ventilated area away from incompatible materials such as organic substances, bases, and reducing agents.
Handling Precautions: Use only in a chemical fume hood. Avoid all contact with skin, eyes, and clothing. Use full PPE and acid-resistant materials.